Aging and Stress
The team focuses on studying the effects of phytohormones and other natural compounds on various parameters related to stress, aging and age-related diseases. The candidate compounds are evaluated in both human primary cell cultures and Caenorhabditis elegans. The expected indications include skin aging, neurodegenerative diseases and sleeping disorders. In the fields of dermatology and cosmetics, we are interested in identification of compounds for the treatment of pigmentation disorders (melanogenesis assays) and psoriasis (keratinocyte differentiation assays). We are also developing cytoprotective compounds with the improved skin bioavailability. Bioinformatic and cheminformatic approaches are used to identify both candidate compounds and molecular targets.
Lab members: Jiří Voller (head), Alena Kadlecová, Václav Mik, Dominika Trofimenková, Tomáš Jirsa

Pharmacognosy and Metabolomics
The group’s research focuses on the discovery of new biologically active metabolites and biomarkers in plants, fungi, bacteria, and other organisms. Natural products represent a rich source of compounds with potential biological activity and continue to serve as an inspiration for the development of a new generation of drugs. We therefore concentrate on their isolation, characterization, and evaluation of activity in various biological systems. Active compounds may function not only as bioactive molecules themselves but also as templates for the synthesis of derivatives with improved properties and higher selectivity.
The identification of active compounds relies on modern metabolomic and bioinformatic methods, with the development of these approaches also being one of our primary research goals. Particular emphasis is placed on the development of computational tools and software for processing LC-QTOF-MS data, enabling the efficient identification of active metabolites. These approaches expand the possibilities of correlation metabolomics and provide deeper insights into the relationship between the chemical structure of natural compounds and their biological effects.
In collaboration with other research groups, we search for compounds with anti-inflammatory, antioxidant, antiproliferative, antimicrobial, and anthelmintic properties. More broadly, we focus on molecules that can influence key cellular processes such as proliferation, apoptosis, angiogenesis, or plant defense mechanisms. These biological activities are essential not only for understanding the functioning of living systems but also for uncovering new opportunities in the treatment of serious diseases, including cancer and chronic inflammatory disorders.
Special attention is devoted to the influence of low-molecular-weight metabolites on plant growth, development, and reproduction. These processes serve as a model system that allows us to study how small bioactive molecules affect complex biological pathways. At the same time, these findings open new avenues for applications in agriculture and biotechnology, where such compounds may contribute to crop protection, increased resilience, or optimized growth.
Lab members: Jiří Grúz (head), Klára Končáková, Andrea Kosinová, Amálie Mikulková
Kinase Inhibitors
Phosphorylation is a universal mechanism for regulating the structure, localization, activity and stability of proteins involved in virtually all cellular processes. It is estimated that 10-50% of proteins can be phosphorylated, often multiple times. Phosphorylation is catalyzed by enzymes called protein kinases and their changes can cause many diseases and disorders including cancers. Genes encoding protein kinases tend to be mutated, amplified and translocated, which in turn significantly alters cell behavior and contributes to tumor transformation. The understanding of these molecular changes has initiated the development of low molecular weight inhibitors of protein kinases, which can be used not only as tools in cell biology, but especially as drugs for cancer.
The development of protein kinase inhibitors as potential antitumor drugs is also taking place in our laboratory. We systematically design, prepare and study compounds based on purine and its isosteric heterocyclic systems, such as pyrazolo[4,3-d]pyrimidines, imidazo[4,5-c]pyridines or imidazo[1,2-c]pyrimidines. Some of our inhibitors show nanomolar activity in both cellular and murine models of cancer. We focus not only on usuall competitive inhibitors, but also on modern modulators of kinase stability based on targeted degradation by PROTAC technologies. The enzymes studied include, in particular, cyclin-dependent kinases (CDKs) and the receptor kinases FLT3 and PDGFR.
Several protein kinase inhibitors are now commonly used as drugs, however, many oncological diseases still rely on conventional cytostatics, the use of which is accompanied by unpleasant side effects. Therefore, we consider this area of research to be very attractive and the development of protein kinase inhibitors on less studied heterocyclic systems is one of our current research intentions.
Team members: Vladimír Kryštof (head), Eva Řezníčková, Radek Jorda, Tomáš Gucký, Miroslav Peřina, Veronika Vojáčková, Petra Krňávková
Structural Biology
The research group has a long-term interest in studying the function of proteins in vitro and in vivo, their structure and binding interactions with ligands (metabolites). The research is focused on two major thematic areas:
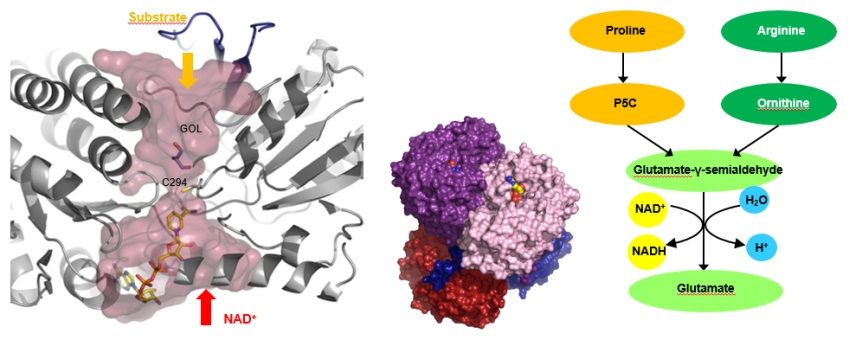
The first topic represents a superfamily of plant and human aldehyde dehydrogenases (ALDHs), which oxidize biogenic and xenobiotic aldehydes using NAD(P)+. Aldehydes are highly reactive compounds and they are generated during the metabolism of carbohydrates, vitamins, biogenic amines, amino acids, steroids and lipids. ALDHs are generally considered as detoxifying enzymes by participating in the adaptive responses to abiotic stress and eliminating aldehydes. In recent years, we have characterized the plant families of ALDH2 (PDB ID: 4PXL, 4PZ2), ALDH7 (PDB ID: 4PXN), ALDH10 (PDB ID: 3IWK, 3IWJ, 4I8P, 4I9B, 4IQ8), ALDH12 (PDB ID: 6D97), and ALDH21 (PDB ID: 5MZ5, 5N5S, 5MZ8) as well as human ALDH7 and ALDH9 (PDB ID: 6QAK, 6QAO, 6QAP).
The second topic comprises enzymes and proteins involved in the metabolism and perception of plant hormones cytokinins, which regulate basic plant processes including cell division, development of meristems, stem, roots, seeds and senescence. These are families of cytokinin oxidase/dehydrogenase (CKO/CKX, PDB ID: 4O95, 3S1C, 3S1D, 3S1E, 3S1F, 4MLA, 4ML8, 4OAL, 3KJM, 2QKN, 2QPM, 5HQX, 5HMR, 6YAO, 6YAQ, 6YAP), nucleoside-N-ribohydrolase (NRH, PDB ID: 4KPN, 4KPO), adenosine kinase (ADK), adenine phosphoribosyltransferase (APRT), and histidine kinase receptors containing the CHASE domain. Modulation of cytokinin levels, through these enzymes, represents an opportunity to improve certain properties of agricultural crops (for example the grain yield or resistance to abiotic stress) in the future.
The research includes techniques for heterologous expression of the gene encoding the studied protein in E. coli, yeast or Arabidopsis thaliana, preparation of overexpressor or knockout lines in the moss Physcomitrella patens in collaboration with the University of Hamburg (Dr. Klaus von Schwartzenberg) or X-ray crystallography in collaboration with IJPB at INRA Versailles (Dr. Pierre Briozzo) and I2BC at the University of Paris-Saclay (Dr. Solange Moréra). In the case of enzymes, the kinetic parameters of substrates and inhibitors are analyzed; the binding parameters between protein and ligand are studied using small-scale thermophoresis (MST). Other techniques include qPCR methods to monitor changes in the gene expression, site-directed mutagenesis is used to study the binding- or active-site of protein, nano differential scanning fluorometry (nanoDSF) is linked to protein stability studies, and CD spectrometry is used to study secondary structures in proteins.
Lab members: David Kopečný (head), Martina Kopečná, Radka Končitíková, David J. Kopečný, Jakub Bělíček